  The atom to atom covalent bonds arrange the silicon atoms in the crystal in an orderly manner. In this way, each atom of a silicon crystal gets 8 electrons in its outermost orbit. 
As there are four electrons in the outer most orbit of a silicon atom, the silicon is a tetravalent element.Įach silicon atom in silicon crystal creates covalent bonds with four neighbouring silicon atoms. The second orbit consists of 8 electrons and the third orbit consists of 4 electrons. Silicon has total 14 electrons in its isolated atom. Silicon requires 1.1 eV for breaking any covalent bond in its crystal and germanium requires This is the main reason for using these two semiconductors most commonly. The silicon and germanium require lower energy to break their covalent bonds in the crystal. Two most commonly used semiconductors are silicon and germanium. There are many semiconductors but few of them are used for electronic circuits. This forms the crystal structure of a semiconductor. In a semiconductor, the atoms are orderly arranged due to atom to atom covalent bonds. #Conductor definition physics freeIn this way, each of them in the crystal will have eight electrons in its outermost orbit.īy forming these covalent bonds, each of the valence electrons in the crystal becomes associated with atoms, hence there will not be any free electron in the crystal in ideal condition. The third orbit consists of 18 electrons and rests 4 electrons are in fourth or outer most orbit.īut in a germanium crystal, each atom shares 4 valence electrons from four neighbouring atoms to fill its outermost orbit with eight electrons. The second orbit consists of 8 electrons. But in an isolated single germanium atom, there are 32 electrons. In germanium crystal, each atom has eight electrons in its last orbit. The figure below shows the covalent bonds formed in a germanium crystal. That means, one covalent bond is created with each of four neighbouring semiconductor atom. Copper is good conductor and glass is insulater.Įach semiconductor atom creates four covalent bonds with four neighbouring atoms in the crystal. Where the resistivity of copper is about 1.7 × 10 – 8 Ω – m in room temperature and resistivity of glass is about 9 × 10 11 Ω – m. 
The resistivity of a semiconductor ranges from 10 – 4 Ω – m to 0. Although a semiconductor has many physical properties, semiconductor name is given to this material because of its moderate electrical conductivity. Because of these four valence electrons, the semiconductor elements do have some special electrical characteristics and properties, which make them useful in using extensively in electronic circuit elements like diodes, transistors, SCRs, etc. The atoms of semiconductor elements have exactly four valence electrons. As the conductivity of those materials lies between good conductors and insulators, and these materials are called semiconductors. 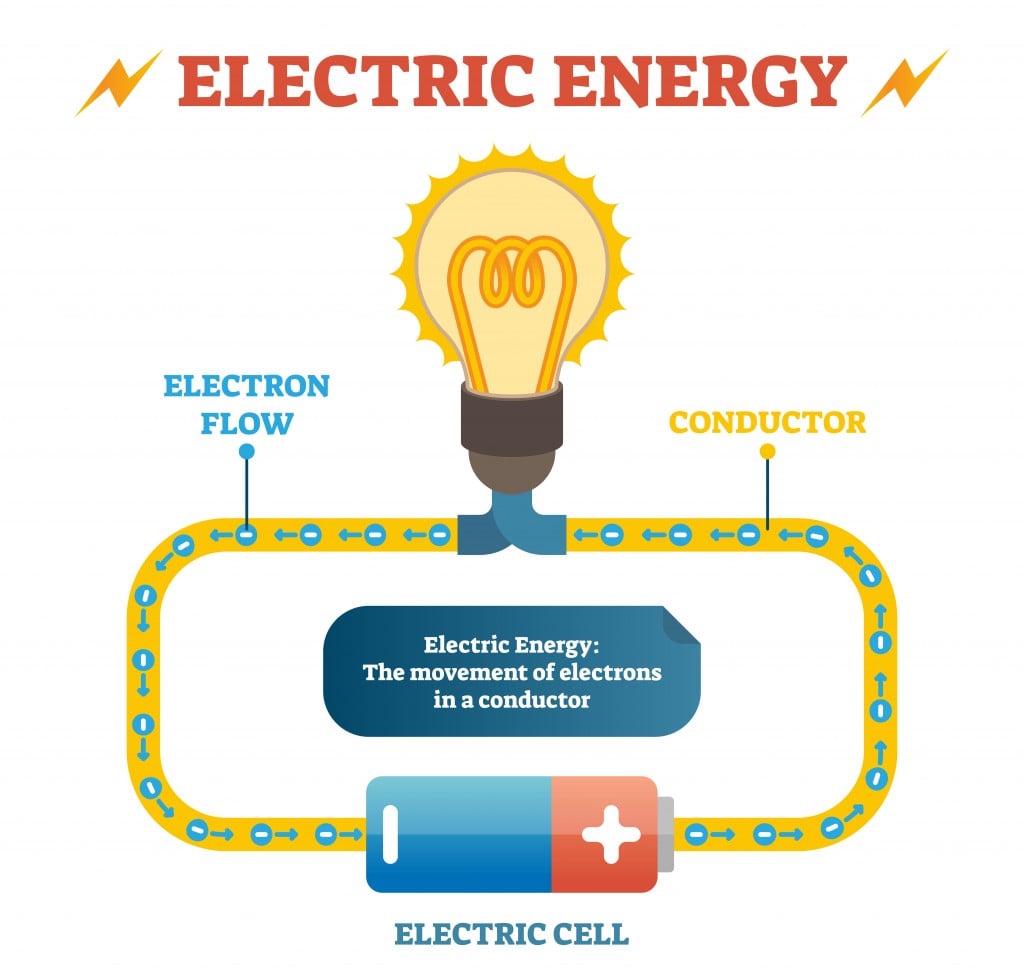
The examples of such materials are germanium, silicon, carbon etc. They have a moderate range of electrical conductivity. Lets see video about ohm law.There are some materials, which have neither good conductivity nor bad conductivity of electricity. In the above figure R=10KΩ, I=5mA, by using ohm’s relation V=IR: We can find voltage by using the relation of ohm’s law V=IR if we know the current and resistance of a circuit.Įxample: Find voltage if the current of 5mA is flowing through the following circuit. I=5mA Calculating voltage using ohmic law Solution: In the above figure R=10 KΩ and V=50 V then from ohm’s law: In order to get current in amperes, you must express the value of voltage and the value of resistance in ohms.Įxample: Calculate the current in the following figure. In the following examples, the formula I=V/R is used. Their voltage-current graph shows not a straight line. or The conductors whose resistance increase or decreases by increasing voltage current and temperature. Non-ohmic conductorsĬonductors or materials which do not obey ohm’s law of ohm are called non-ohmic conductors or materials. Some of the examples of ohmic conductors are: Current- voltage graphs of these materials show a straight line. Metals mostly are ohmic conductors, their resistance remains constant by increasing voltage current and temperature. ohms law triangle difference between ohmic and non-ohmic conductorsĬonductors that obey ohm’s law are called ohmic conductors in other words materials whose resistance is constant are called ohmic conductors. ohm’s law equationĮquation (1) is the ohm law equation in which “V” is voltage “I” is the current and “R” is the resistance. V has applied voltage in volts and I is current in amperes. The value of the resistance depends upon the nature, dimensions, and physical state of the conductor. Where R, the constant of proportionality is called the resistance of the conductor. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
